Equation for atomic mass number9/2/2023 
Now, you can calculate the atomic weight of carbon in the sample.įor this sample, the atomic weight of carbon is 12.0107. The relative atomic mass of carbon-12 is defined as exactly 12 and the relative atomic mass of carbon-13 isġ3.00335. To calculate the atomic weight of carbon, you must also know the relative atomic masses of each of carbon's stable isotopes. if we see the hydrogen atom which contains one. To calculate the numbers of subatomic particles. These abundances are very similar to those that can be Simply as we can better understand the atomic number by taking an example i.e. Example 2: Oxygen atom has 8 protons and 8 neutrons. Assume that in the sample, the abundance of carbon-12 is 0.9893 and the abundance of carbon-13 is 0.0107. Example 1: The hydrogen atom doesn’t have a neutron but it has 1 proton, so the mass number of hydrogen is 1. Now you know that atomic number number of protons, and mass number number of protons + number of neutrons. You also need to know the relative abundance of each of the isotopes. Questions Tips & Thanks Want to join the conversation Vica Kelly 8 years ago This is probably a very stupid Question, but I have to ask it. Question Balance the nuclear equation by giving the mass number, atomic number, and element symbol for the missing species. Change each percent abundance into decimal form by dividing by 100. 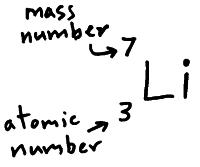
Step 1: List the known and unknown quantities and plan the problem. Carbon has two stable isotopes: carbon-12 and carbon-13. Use the atomic masses of each of the two isotopes of chlorine along with their respective percent abundances to calculate the average atomic mass of chlorine. First, you need to know the number of stable isotopes of carbon. In order to calculate the molecular weight of one water molecule, we add the contributions from each atom that is, 2(1) + 1(16) 18 grams/mole. Recall the information that is required to calculate an atomic weight. Now, use the formula below to determine the atomic weight of carbon in a particular sample without using the learning tool, and then check your answer. Together, the number of protons and the number of neutrons determine an element's mass number: mass number protons + neutrons. The isotopic abundances are, by default, set to the approximate abundances that areįound in nature. Forms of the same atom that differ only in their number of neutrons are called isotopes. Click on the large element symbol to pull up the Periodic Table and select carbon. To determine relative atomic mass, we simply multiply each isotopic mass by its abundance, add all the values together and divide the total value by 100 percent. For now, use the Atomic WeightĬalculator to find the atomic weight of carbon in a sample. The atomic mass (number at the bottom) is the amount of protons and neutrons added together. So if an element has an atomic number of 5, you know that it has 5 protons and 5 electrons. The short answer is that you can find a power-law fit ($1.Open the Atomic Weight Calculator to explore the relationship between the isotopic abundances of an element and the element's atomic weight. The atomic number (number at the top) is the amount of protons and the amount of electrons.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
